Procedures
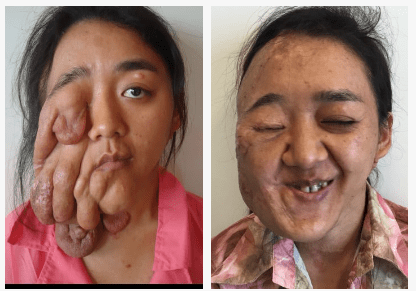
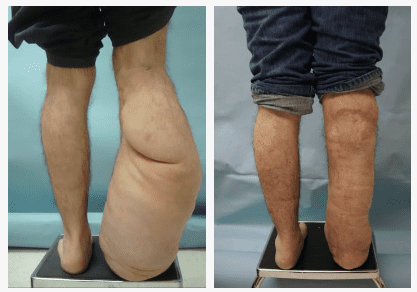
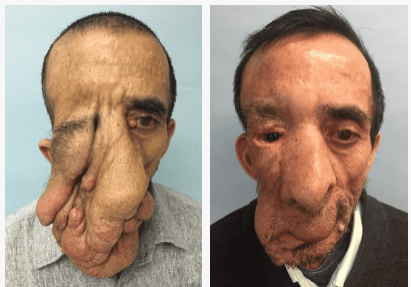
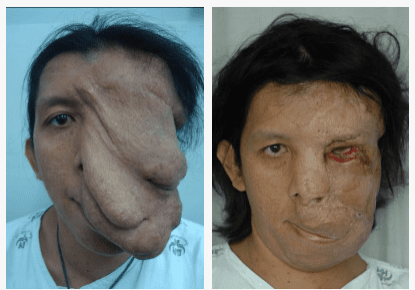
Cases in Neurofibromatosis Treatment
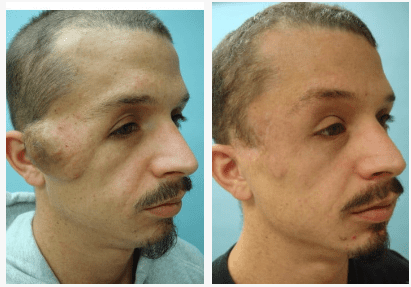
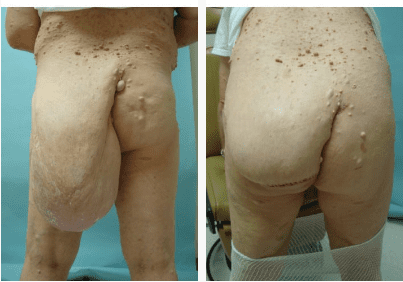
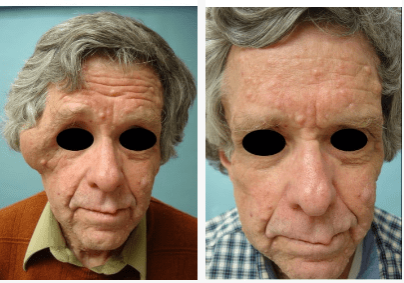
Click Images below for a more detailed view. Individual results may vary.
SURGERY OF NEUROFIBROMATOSIS
HISTORY
Neurofibromatosis, or, what we commonly call NF1, is an autosomal dominant genetic condition of chromosome #17 which leads to formation of tumors of skin and all other parts of the body. Approximately 1 in 3000 persons are affected. One half of all cases are
spontaneous mutations, the rest of patients having inherited the gene from a relative.
The original descriptions of the disease were by Von Recklinghausen in the 1860’s. The lesions he described were mainly of the skin, which represent the majority of patients with NF1. Other NF1 tumors which can emerge in both skin and deeper areas of the body are called plexiform tumors. These tumors, once they have appeared, have a strong tendency to grow
unpredictably, similar to other solid tumors. Plexiform tumors also have the ability (5-10%) to transform into a malignant form of NF1 called Malignant Peripheral Nerve Sheath Tumor (MPNST). These tumors present the greatest risks of pain and damage to a patient’s normal
tissue and functions.
SURGERY
The history of surgery for resection of cutaneous tumors is mostly successful since they are easily accessible and small, although recurrence of tumor has been a problem for many patients. Surgery of plexiform tumors has a history of significant blood loss, high recurrence of tumors and unintended but permanent injury to normal nerves and tissues.
This history has nurtured a wariness by pediatricians and surgeons to postpone surgical attempts at removal until after puberty or until serious morbidity from the tumors is present. This default strategy results in significant morbidity and suffering for patients since unchecked tumor growth is the usual result of no surgery.
Dr. McKinnon’s Experience
Over a 40 year experience of treating patients with NF1, I have observed that “radical” resection of plexiform NF tumors has resulted in permanent “non-recurrence” of nearly all tumors. This includes even tumors over 50 pounds and some MPNST tumors. In children, who have enormous growth spurts until the end of puberty, there is no certainty of permanent surgical results. However the successful logic gleaned from radical surgery in adults has been successfully applied to a large pediatric population, and without facial mutilation, even if surgery may need to be repeated during growth. This success also depends on preservation of normal motor nerves and other normal tissue during the resection of NF tumors.
ANATOMY
CRANIOFACIAL
Most facial and frontotemporal scalp tumors derive from sensory branches of cranial nerve V. The ophthalmic division of the trigeminal nerve includes some sensory nerves that are within the musculofascial cone (annulus of Zinn) of the orbit. This area presents special risks of injury to the optic nerve and the extraocular muscles and should be avoided in surgery.
Orbital NF is often (even from birth) accompanied by a defect of the sphenoid greater wing. This defect may permit herniation of the temporal lobe of the brain into the orbit and subsequent orbital dystopia, pulsation exophthalmos or enophthalmos, and pressure on the globe and optic nerve. Destruction of bone, ligaments, fascia, muscle and skin may also be present.
Tumor confined to the optic nerve (optic glioma) deserves neurosurgical management. It is important (and an original observation by Dr. McKinnon) to understand that motor nerves are not intrinsically involved with NF tumor. NF tumors of the mandible, parotid gland, ear and temporal scalp develop largely from the mandibular division of the trigeminal nerve, including the auriculotemporal nerve. Tumors of the neck and posterior scalp derive from upper cervical sensory nerves (C1-C4), including theoccipital nerves. Lower cervical nerves (C4-C8, sensory and motor) constitute most of the brachial plexus.
TRUNK AND EXTREMITY
Plexiform NF can exist anywhere from the spinal cord to the sensory nerve terminus in the skin, bone or viscera. Plexiform tumors most commonly appear between the deep fascia and the skin but may involve and destroy skin, muscles, bones, joints, and visceral organs.
Careful dissection proximal to the tumor mass (towards their CNS origin) often reveals the specific sensory nerve tumor origin. Large truncal lesions over time can develop paraspinal hypervascularity. This can resemble an arteriovenous malformation. Scoliosis of the spine is common with posterior trunk lesions, and limb hyperplasia is common with extremity lesions.
PATHOGENESIS
NF is an autosomal dominant condition which can be attributed to a defect on chromosome 17, but mosaicism can also occur. The consequential lack of neurofibromin (a tumor inhibitor) permits growth of myelinated axonal tumors in any bodily location. Tumor types are cutaneous or plexiform. So-called plexiform tumors are solid tumors.
They have an obligatory angiogenic influence, similar to other solid tumors. They express multiple angiogenic and neurogenic factors. Puberty, pregnancy and childhood growth spurts can exacerbate their growth. Tumor vascularity increases proportionate to tumor growth. Transformation of benign NF tumors into MPNST can occur in plexiform tumors. This transformation may have a direct correlation to numerical tumor cell replication events (i.e., the more cells replicate, the greater chance they have of producing malignant cells).
Tumor vessels in NF are pathological arteries that reveal absent media and abnormally thin adventitia. Established tumors can also exhibit vascular lakes and/or nests of abnormal vessels resembling a congenital vascular malformation.
NATURAL HISTORY
Neurofibromas, once established as plexiform tumors, do not regress spontaneously. Their growth is unpredictable and non- regular, including possible onset throughout a patient’s life of new lesions. Malignant transformation is more common in truncal/extremity lesions but is also unpredictable. Cafe au lait cutaneous spots do not necessarily signal the presence of plexiform tumors, although they themselves represent microscopic tumor. If present at birth, plexiform tumors may have already inflicted defective tissue development of the newborn.
Eventual destruction by tumor of adjacent skin, bone and other tissue should be expected without intervention. Hyperplasia and/or fragility of bone occurs in extremity lesions. Craniofacial tumors can also produce osteoclastic or osteoblastic pathology, likely as a consequence of tumor growth rate. In other words, slow growing tumors increase local growth, fast growing tumors destroy local tissues.
Tumors involving the sphenoid and orbit can cause progressive orbital dystopia, blepharoptosis and blindness. Paraspinal tumors inflict progressive scoliosis on the growing child.
PATIENT HISTORY AND PHYSICAL FINDINGS
The myriad presentations of NF tumors should not deter accurate diagnosis, even in infancy, by the observant neurologist or experienced surgeon. If doubt exists of the diagnosis, a biopsy should be performed with exception at least of the optic nerve and brain.
Physical findings are too numerous to justify an inclusive list, but palpable tumor, pain or confirmatory imaging should precede surgical intervention in virtually all cases.
IMAGING
Pediatric patients with suspicion of plexiform tumor should have an MRI study, preferably with and without contrast. MRI of the CNS is appropriate as an early screening measure for all NF patients, even those without known tumors. T2 and STIR-weighted images usually give the clearest depiction of NF tumor.
MRI, especially with higher Tesla power, can frequently reveal the specific cranial or spinal nerve involved with tumor. This information (nerve mapping) should be sought out with the aid of a neuroradiologist.
High-resolution CT scans should only be ordered when and if the patient with skeletal destruction or high risk of skeletal pathology is being evaluated preoperatively.
DIFFERENTIAL DIAGNOSIS
NF tumors have been mistaken for vascular malformations, particularly by their hypervascular imaging on CT or MRI. Hyperpigmentation of NF lesions has been confused by the inexperienced physician with
congenital nevi. A biopsy can readily produce the correct diagnosis.
Most patients, even infants, with NF will present with at least three or more cutaneous lesions and/or cafe au lait lesions, axillary freckling, ocular Lisch nodules or a subcutaneous plexiform mass.
Congenital ptosis can usually be differentiated from NF-derived ptosis by detection of an orbital mass, presence of enophthalmos or exophthalmos, and by MRI findings.
Rapid increase of ptosis may be associated with orbital malignancy such as Rhabdomyoscarcoma, for which immediate incisional or excisional biopsy is required.
Diagnosis of MPNST or other orbital malignancies cannot be reliably made by symptoms, by imaging or by clinical examination.
NONOPERATIVE MANAGEMENT
This default management decision is historically common but deserves renewed challenge because it is controversial. Observation of growing NF tumors is patently non therapeutic yet may be justified if surgery is high risk, the patient has significant comobidity, or the tumor has not reached a level of irreversible morbidity to vital structures. The surgeon and his or her medical colleagues should be prepared to intervene to prevent irreversible morbidity, even in young patients.
SURGICAL MANAGEMENT
An accurate diagnosis is the sine qua non of surgical management. With plexiform NF this should include determination of the tumor nerve origin(s), the degree of vascularity, bony deformity, proximity to other vital anatomy, and degree of skin destruction. In the craniofacial tumors, accurate diagnosis also should determine presence of brain herniation into the orbit (or elsewhere), orbital dystopia, ptosis versus pseudoptosis, ophthalmic pathology and airway and carotid/jugular risks.
Because all NF1 tumors are caused by pathologic sensory nerves, either of brain or spinal cord origin, if a total interruption of the responsible sensory nerve(s) can be achieved (e.g. a Sunderland type V injury), permanent non-regrowth of tumor may also be achieved.
Thus, surgical strategy should aim to:
Radically resect the plexiform tumor and identify and amputate the responsible sensory nerve(s) proximal to the tumor. Dissect and preserve any motor nerves in the tumor proximity.
Undertake tumor resection even in the pediatric patient because progressive tumor growth over time equates to more complex surgery, higher risk of morbidity to normal adjacent structures and function, and possibly more risk of malignant transformation of previously benign tumors.
All techniques are subservient to the principles of reconstructive surgery. A knowledge of and adherence to principles permit the surgeon to methodically approach virtually any surgical problem to arrive at an organized plan. The reconstructive surgeon’s “creed” has been summarized by Millard: “Know the ideal beautiful normal. Diagnose what is present, what is diseased, displaced or distorted, and what is in excess. Then, guided by the normal in your mind’s eye, utilize what you have to make what you want—and when possible go for even better than what would have been!”
No clinical condition presents to the surgeon a more variable array of pathology than Neurofibromatosis. The NF surgeon needs, therefore, to first make a strategy from application of principles. When the principles (such as making an accurate diagnosis, establishing priorities, making a plan and backup plan, replacing lost tissue in kind, etc.) are applied, the surgical techniques will devolve naturally. The surgeon, much like his or her military counterpart, eludicated the goals and strategy of a military-type campaign before determining the specific technique(s) required.
Suspicion of MPNST diagnosis either by clinical findings or PET scan should presage a surgical strategy of en bloc resection and marginal biopsies to ensure complete resection.
PREOPERATIVE PLANNING
Pertinent consultations with other specialists should be completed well prior to surgery, if feasible. Adequate warning to blood bank and ICU is made.
APPROACH
Orbital tumors are approached via coronal, superior lid crease and transconjunctival incisionsas needed. Inferior orbital nerve (maxillary) tumors are accessed via a lower eyelid incision as in orbital floor exploration or via a nasojugal incision.
The surgeon should anticipate repeat procedures (i.e., a subciliary incision is always a higher risk for ectropion). Posterior trunk tumors should be accessed via incisions that can resect not only the palpable tumor but also any feeding paraspinal sensory nerves.
GENERAL PRINCIPLES
The first priority of surgery in the child with NF is resection of tumor and reconstruction of bony defects. Large tumor resection can be aided by cautery devices such as LigaSure, but the surgeon should also be facile with vascular surgical techniques.
Autogenous bone reconstruction is superior in virtually all cases to alloplastic reconstruction and homografts, especially in the growing child.
Because resection of cranial nerve V tumors into the cavernous sinus is dangerous (and some tumor will necessarily remain there), reconstruction of the sphenoid defect should be undertaken with a combination of bone and metallic plate/mesh to ensure permanence of brain-orbit separation (even in the enucleated patient).
Bone should face the orbit to allow possible attachment of the levator muscle origin .
Early ptosis correction is important to avoid amblyopia, but definitive ptosis correction should be performed when the child can tolerate a non- general anesthetic for refinement of the correction by patient participation and the tumor is not significantly present.
When approaching a large plexiform tumor, the surgeon should begin dissection in normal tissue and proceed to identify the plane at the tumor margin. Motor nerves that are within the tumor field should be identified by direct nerve stimulation and reconfirmed to be normal at conclusion of surgery.